IMMUNOMONITORING AND CHARACTERIZATION FOR PROTEIN-BASED DRUG DEVELOPMENT
Accelerated Protein Drug Development
Meet ABL’s immunobiology team at PEGS to discuss ABL’s immunomonitoring, efficacy, and protein characterization services!
ABL will be attending the PEGS: The Essential Protein Engineering Summit from April 30 – May 4 at the Seaport World Trade Center in Boston, MA. Contact us today or visit us at booth #200 to see how ABL can support the design, characterization, immune monitoring and safety of your protein-based vaccine or therapeutic!
As a CRO and CMO with over 50 years of experience partnering with clients to meet their product development goals, ABL’s Immunobiology team provides contract research services from preclinical development to the commercial market. Top analyses ABL scientists can perform to evaluate your protein product:
- Binding Kinetics, Antigen Selection, Isotyping, and Fc Receptor Function via Biolayer Interferometry (ForteBio Octet Red96)
- Linear Epitope Mapping with Pepscan
- Multiplexed Cytokine/Biomarker Detection and Monitoring (Luminex 200, MesoScale Diagnostics QuickPlex SQ120)
- Ultra-Sensitive Biomarker Detection Assays (Singulex Erenna, Quanterix Simoa, Bio-Rad QX200 Droplet Digital PCR System)
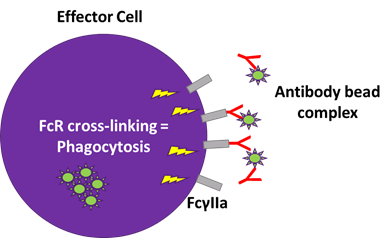
- Fc effector function with Antibody Dependent Cell-mediated Cytotoxicity (ADCC) and Antibody Dependent Cellular Phagocytosis (ADCP)
- Pharmacodynamic Evaluation via Flow Cytometry-Based Receptor Occupancy Assay
- Immunogenicity and Anti-drug Antibody Assays

We can also assist in the process development and GMP manufacturing of your protein with our manufacturing services.
Contact us now to speak with one of our experts:



